Properties of Nail Polish
Chemical Formula of nail polish
The chemical formula of nail polish is not available to the public because the precise formula is a trade secret.
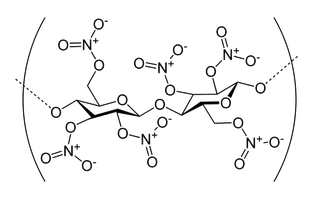
Nail polish is a fairly complex substance made up of many different chemicals. However, almost all types of nail polish contain nitrocellulose, also known as guncotton.
Nail polish is a fairly complex substance made up of many different chemicals. However, almost all types of nail polish contain nitrocellulose, also known as guncotton.
Nitrocellulose is what makes nail polish dry properly and what forms the smooth surface of the polish. Nail polish needs to dry in a film and dry quickly, but it can't dry so quickly that only the top layer fully dries. Nitrocellulose helps the nail polish to dry quickly, yet also slowly enough so that none of the layers remain wet. It is also used because of its particular durability.
Other components of nail polish include resins, plasticizers, pigments, and solvents.
The most common resin is tosylamide/formaldehyde resin. It changes the nitrocellulose polymers to create a tough, shiny, and long-lasting shell. This makes the nail polish endure for a lengthy period of time while maintaining its glossiness. The resin also makes the varnish stick to the nail better.
Dibutyl phthalate is another ingredient in nail polish. It is mainly a plasticizer that helps the nail polish maintain its flexibility and durability over time. Plasticizers also prevent chipping and cracking.
The pigments in the nail polish are different depending on the colour of the nail polish. In some types of varnish, there are ultraviolet filters, to make sure the sun does not damage the colour of the polish. Sometimes, nail polish will contain micas, little pieces of reflective materials, and pearl essence (guanine) to give the varnish more shimmer and shine.
Butyl acetate and ethyl acetate are some of the most common solvents. The solvent must hold the materials in the nail polish until the varnish is applied to the nail, then evaporate to leave a colourful finish. It must dry quickly to increase the ease of the nail polish application. Bothe butyl acetate and ethyl acetate can be used as nail polish removers. Toluene and formaldehyde can also be used as solvents (formaldehyde is no longer very common, but it was previously found in many varnishes).
Other components of nail polish include resins, plasticizers, pigments, and solvents.
The most common resin is tosylamide/formaldehyde resin. It changes the nitrocellulose polymers to create a tough, shiny, and long-lasting shell. This makes the nail polish endure for a lengthy period of time while maintaining its glossiness. The resin also makes the varnish stick to the nail better.
Dibutyl phthalate is another ingredient in nail polish. It is mainly a plasticizer that helps the nail polish maintain its flexibility and durability over time. Plasticizers also prevent chipping and cracking.
The pigments in the nail polish are different depending on the colour of the nail polish. In some types of varnish, there are ultraviolet filters, to make sure the sun does not damage the colour of the polish. Sometimes, nail polish will contain micas, little pieces of reflective materials, and pearl essence (guanine) to give the varnish more shimmer and shine.
Butyl acetate and ethyl acetate are some of the most common solvents. The solvent must hold the materials in the nail polish until the varnish is applied to the nail, then evaporate to leave a colourful finish. It must dry quickly to increase the ease of the nail polish application. Bothe butyl acetate and ethyl acetate can be used as nail polish removers. Toluene and formaldehyde can also be used as solvents (formaldehyde is no longer very common, but it was previously found in many varnishes).
Physical and Chemical Properties of nail polish
The physical properties of nail polish include:
The chemical properties of nail polish include:
- Quick drying time (yet slow enough for all the layers to dry equally)
- Liquid form until applied to nails
- Unless specified as "matte", shiny appearance
- Durable, difficult to chip
The chemical properties of nail polish include:
- Not reactive with water
- Very flammable
- Creates a hard film when exposed to air for a prolonged period of time